Chapter 4.35
4.35 - Legume Rhizobia Inoculation Using Nodule Crushing
Katherine Stevenson,University of Guelph, Canada
Suggested citation for this chapter.
Stevenson ,K. (2025) Legume Rhizobia Inoculation Using Nodule Crushing. In Farmpedia, The Encyclopedia for Small Scale Farmers. Editor, M.N. Raizada,University of Guelph, Canada. http://www.farmpedia.org
What is it?
Nitrogen fertilizer is an essential and often limiting requirement for plant growth. Nitrogen is needed to make DNA, protein and chlorophyll (Pudasaini & Raizada, 2024). Although nitrogen is abundant as a gas (N₂) in Earth's atmosphere, it is inaccessible to plants due to the strong triple bond holding the molecule together. To be usable, nitrogen gas must be broken apart and become available in a solid form (“fixed”).
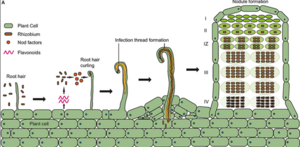
Certain bacteria produce the enzyme nitrogenase, which breaks the N₂ triple bond to create ammonia (NH₃) which converts to ammonium (NH₄) — a common nitrogen fertilizer (Andrews & Andrews, 2017). This process is called biological nitrogen fixation. Plants cannot produce nitrogenase themselves because it requires a low oxygen environment, whereas most plant tissues have high concentrations of oxygen (Hemida Abd-Alla et al., 2023). However, bacteria in the genus Rhizobium have co-evolved with legume plants (Leguminosae) to form a symbiotic relationship. Legume crops include common bean, soybean, chickpea, lentil, fava bean and more. Legume roots release natural chemicals (flavonoids) that attract local rhizobia around the roots (Li et al., 2021). The bacteria then enter the roots, multiply, and stimulate the production of small root organs called nodules (see Figure 1 and Figure 2) where the bacteria make the natural ammonia fertilizer (Deaker et al., 2004). In this reciprocal relationship, plants provide sugars to the bacteria, which in turn supply nitrogen fertilizer, enriching soil health and enhancing plant growth (Andrews & Andrews, 2017).
Figure 1. Simplified step by step diagram of interaction between soil rhizobia bacteria and legume roots during infection. The diagram displays the development of an infection thread – the most common mode of infection. The rightmost structure on the image shows a mature nodule containing multiple functional zones (Wang et al., 2018)
The challenge is that some soils do not contain a compatible or effective rhizobia bacteria, especially if a crop species is new to a region (Pudasaini et al., 2023). One strategy to promote nitrogen fixation is to coat seeds or spray fields with an improved and/or compatible bacterial inoculant; this has been shown to increase nodulation and can improve legume yields by up to 25% (Chibeba et al., 2018; Deaker et al., 2004). Rhizobia bacteria can be purchased commercially; however such bacteria may not be available in remote areas and require costly infrastructure and refrigeration, making it inaccessible to many small-scale farmers, especially in Sub-Saharan Africa (Pudasaini & Raizada, 2024).
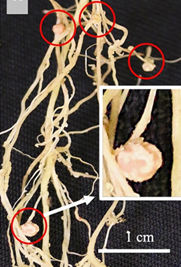
After improved rhizobia have become available in a region, a simpler alternative is to use the first-generation inoculated plants to spread the rhizobia to nearby farms by crushing their root nodules onto new seeds (see Figure 3). Alternatively, farmers can select the best nodules from their own fields to crush onto seeds. In this technique, farmers first visually select the healthiest legume plants as a source of nodules, then select the largest and pinkest root nodules from these plants, crush the nodules using local tools and coat them onto seeds using a locally available sticky agent such as Gum Arabic or molasses (Pudasaini et al., 2023). This technique is called ‘nodule crushing’ (Pudasaini et al., 2023). When planted, the inoculated seeds provide an immediate source of effective rhizobia, improving nodulation, plant growth, and reproduction, especially in a field’s first season (Pudasaini et al., 2023).
Why Legume Crops?
Firstly, this strategy only works with legume crops because of their unique symbiosis with the nitrogen-fixing bacteria rhizobia. Since the bacteria form nodules in the roots of legumes, the source material (i.e. nodules) required for inoculation is readily available. In every region of the world, there is a traditional legume crop species. This makes the technique versatile and applicable to small scale farmers worldwide.
As an added benefit, legumes are very nutritious staple crops. Deficiency in some of the building blocks of protein, called amino acids, is a serious but common issue that impacts one billion people worldwide, predominantly in rural areas especially where meat consumption is low (Pudasanini & Raizada, 2024). Nitrogen is a key component for amino acid synthesis, and thus supplementing fields with nitrogen has been shown to improve the protein content of seeds and leaves (Britz et al., 2023). When combined with a cereal grain such as maize, wheat, or rice, a legume-based meal provides all 9 essential amino acids, making them a very nutritionally valuable crop family (Chibeba et al., 2018).
Synthetic nitrogen fertilizer is an expensive and environmentally disruptive solution which contributes to climate change (Deaker et al., 2004). However, most legume crops do not need this supplemental fertilizer to produce a crop that is a good source of protein (Chibeba et al., 2018). Soybeans can source 84% of their required nitrogen through biological nitrogen fixation, but when synthetic nitrogen is applied this percentage drops significantly (Gelfand & Robertson, 2014). Like other legumes, their levels of biological nitrogen fixation is highest when no additional nitrogen is applied. A 2008 review by Herridge et al., found that actual levels of nitrogen fixation in soybeans vary depending on environmental and genetic factors, but the average is 227 kg/ha. In addition to supplying nitrogen to the plant biomass, soil nitrogen reserves are also replenished by biological nitrogen fixation (Herridge et al., 2008).
Benefits of Seed Inoculation
Timing of Sowing & Harvesting
With traditional planting, legumes rely on the rhizobia bacteria already present in the soil to colonize their roots for nitrogen fixation (Li et al., 2021). This is because, without inoculation by farmers, rhizobia are not inherited to future generations of seeds. In soils degraded by erosion, leaching, salt, or synthetic fertilizers, or in fields that have never grown legumes, nitrogen-fixing bacteria may be ineffective, scarce, or entirely absent. In these cases, nodulation will be low, and the potential benefit gained from biological nitrogen fixation is extremely reduced (Deaker et al., 2004).
The primary benefit obtained from the nodule crushing technique is the improvement in legume yield. Inoculating the rhizobia directly onto seeds improves nodulation and nitrogen fixation (Pudasanini & Raizada, 2024). This is important, because currently, there is a significant gap in the yield of legumes between Africa (1.1 t/ha) and North America (2.6 t/ha) contributed to by low nitrogen soils. Nodule crushing and seed inoculation represent an environmentally and economically sustainable way to begin to address this challenge. A review by Deaker et al. (2004) found that 59% of variability in legume yield was caused by the number of rhizobia present in the soils at times of sowing, while inoculation of soils directly increased yield by 25% alone. Introducing this pre-planting intervention may increase yields of legumes where they are historically low, fighting chronic food insecurity.
Furthermore, it is hoped that farmers can use repeated cycles of nodule crushing from the healthiest plants and nodules to improve the rhizobia bacteria. In trials of nodule crushing with common bean, nodule count, shoot yield, and chlorophyll improved after four cycles of selection, crushing, and inoculation (Pudasaini, 2024).
How to Implement?
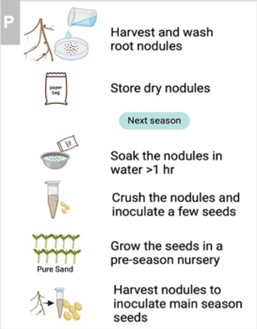
See Figure 3 (above) for visual instructions To inoculate a small legume field (<1 ha) with rhizobia:
1. Select the 10 largest, healthiest looking plants near the end of the growing season, but not too late, because otherwise the nodules will be dead:
- Greenest plants
- Largest plants
2. Dig up the plants and wash the roots with water.
3. Collect the 10 healthiest nodules from each plant:
- Large nodules
- Pink colour nodules (See Figure 4)
- If the nodules are too small, then purchase an inexpensive magnifying glass from a local store to view them.
4. Dry nodules at room temperature, away from the sun, for 2-3 days. They will shrink in size.
5. Store dried nodules in a cool dark place until next season, protected in a bag or jar:
- Different species nodules should be kept separate and labelled, because only nodules of the same crop species are compatible.
6. The nodules need to be multiplied at the start of the growing season. Starting 4-6 weeks before the start of the main growing season, crush the nodules onto 10-50 plants which may be planted in a small nursery using irrigation. These nursery plants should be planted in pots or bags with sand and a little manure, but not soil, because the soil may contain non-optimal rhizobia bacteria which will compete with the bacteria that you are trying to introduce. The nodule crushing technique is explained below:
7. Fill a small pot of water with freshwater, add a small spoon or fork, then boil the water for 10 minutes (with the spoon/fork), and then let it cool completely. Wash your hands with soap, and then remove the spoon/fork and place it on a clean cloth until the next step. Immediately, soak the dried nodules in this water for ~1 hour. You can watch the nodules become larger in size. (See Figure 5)
8. Remove most of the water from the pot, except a small amount, but be careful to keep the nodules in the pot. Then crush nodules in the pot using the boiled spoon/fork.
9. Mix nodule solution with a sticky substance and 10-50 seeds:
- Sticky substance should be non-toxic
- Molasses, gum Arabic, etc.
10. Grow seeds in pots or bags in the pre-season nursery in sand with a little manure (but not soil). Purpose: to multiply nodules for sale or planting
11. After 4-6 weeks, harvest all the healthy nodules from the nursery plants and use these for crushing onto seeds for the main field.
12. Plant seeds in the field at the typical planting date.
13. At the end of the season, repeat the process by selecting the healthiest plants and harvest the largest, pink nodules.
Figure 4. Pink root nodules indicating active nitrogen fixation. Pink interior in the nodules indicates the presence of leghemoglobin, a protein present when nitrogen fixing symbiosis is present (Pudasaini & Raizada, 2024).
Advantages of Nodule Crushing and Seed Inoculation
Legume seed inoculation using nodule crushing has numerous advantages over commercial field inoculants, especially for small-scale farmers with limited resources (Pudasaini et al., 2023). Using this decentralized strategy puts power and autonomy directly in the hands of farmers instead of making them reliant on large companies or governments for supplying inoculants. Commercial inoculant is mass produced, meaning it is generic and may not provide the most compatible strain of rhizobia; or it may be contaminated by other microbes altogether. Keeping control in the hands of farmers allows them to be self-sufficient in producing inoculant (Pudasaini et al., 2023).
Through yearly selection of the healthiest plants and the pinkest nodules, farmers may be able to artificially select the most compatible strain of rhizobia for their specific cultivar. This should allow for selection against cheaters – non-nitrogen-fixing bacteria that inhabit root nodules to gain sugars from the plant – improving the efficiency of growth (Pudasaini et al., 2023). Crushing of nodules passes on the entire microbiome within the nodule, also preserving the genetic diversity of the bacteria selected (Pudasaini et al., 2023). Each round of selection and inoculation is a potential opportunity for an increase in compatibility and effective nitrogen fixation, allowing for continual improvement each year. This strategy may also allow for the co-evolution of rhizobia strains and legume landraces, to maximize success of symbiosis in a specific region. Additionally, selection of healthy plants, seeds, and nodules at each step may greatly reduces the risk of diseases being spread intergenerationally (Pudasaini & Raizada, 2024).
Farmers who participate in this strategy may also be able to sell their inoculated seeds and share benefits with others in their community. The sale of either nodules or inoculated seeds will promote biodiversity and the exchange of genetics between farmers, protecting soil biodiversity in these agricultural fields. Their sale is also an opportunity for additional income generation for farmers. This is a good opportunity for women to make money, as the labour of female farmers is often undercompensated (Pudasaini et al., 2023). Having a decentralized distribution framework means each stage of economic interaction provides benefits to numerous local individuals involved in sales and distribution. Additionally, having decentralized inoculant production creates the opportunity for a diversity rhizobia to accompany associated diversity in crop species. In isolated locations, staple crops are often specific landraces that have arisen from years of artificial selection. Sourcing microbial inoculants from other farmers in the region would likely prove to be more successful than the inaccessible commercial alternative.
Critical Analysis
While nodule crushing and seed inoculation offer a low-cost, decentralized alternative to commercial inoculants, the strategy has some limitations. One concern is the potential impact on rhizobia diversity. As farmers selectively save strains from the most successful nodules, genetic diversity within the microbial population may decline over time. This could reduce adaptability to changing soil conditions and limit the long-term resilience of communities as climate changes (Pudasaini et al., 2023). Additionally, widespread exchange of inoculated seeds may unintentionally introduce non-native rhizobia strains, potentially disrupting local soil microbiomes.
Another drawback is that this strategy alone may not match the effectiveness of synthetic nitrogen fertilizers. While rhizobia can improve nitrogen availability, the total amount fixed is limited by environmental conditions, soil health, and legume efficiency. In contrast, synthetic fertilizers provide an immediate and predictable nitrogen source, supporting higher yields, particularly in nutrient-depleted soils. This means that while inoculation enhances sustainability and accessibility, it may not fully replace synthetic fertilizers for maximizing crop productivity particularly in less effective nitrogen-fixing crops such as common bean (Phaseolus vulgaris) which is an important crop in East Africa and Latin America (Pudasaini et al., 2023).
It must also be considered that this strategy is still currently in its pilot stage. This means that while it has been effective in a research environment, success under farmers’ fields has not been tested and thus can not be guaranteed. In a research environment, sterile materials and carefully regulated variables ensure consistent results. However, on small scale farms, numerous environmental uncertainties—such as soil variability, microbial competition, and climate fluctuations—could reduce its overall effectiveness (Pudasaini et al., 2023). Since this strategy has not yet been implemented by small-scale farmers or tested under uncontrolled conditions, both the methodology and practical recommendations may need to be adjusted based on field results. There is a strong need for on-farm testing and further research to determine the practical performance of this strategy.
Practical Links & Resources to get Started
Root nodule formation:
https://www.youtube.com/watch?v=O8WbVzXdI14
Nitrogen fixation:
https://www.youtube.com/watch?v=p3uJHrvPeKo
Detailed instructions and further reading:
SAKbooks.com. Chapter 5 Lessons 21-24:
5.21: Legume Nodules
5.22: Benefits of rhizobia
5.23: Nodule assessment
5.24: Seed inoculationhttps://sakbooks.com/south-asia-english-version-all-chapters/
https://sakbooks.com/east-southeast-asia-english-version/
https://sakbooks.com/sub-saharan-africa-caribbean-english-captions/
https://sakbooks.com/north-africa-middle-east-english-version/
References
1. Andrews, M., & Andrews, M. E. (2017). Specificity in legume-rhizobia symbioses. International Journal of Molecular Sciences, 18(4), 705. https://doi.org/10.20944/preprints201608.0005.v1
2. Britz, E., Cyster, L., Samuels, I., Cupido, C., Masemola, L., Ngcobo, N., Manganyi, F., & Müller, F. (2023). Nitrogen fertilization increases the growth and nutritional quality of the forage legume, Calobota sericea – a preliminary investigation. Heliyon, 9(2), e13535. https://doi.org/10.1016/j.heliyon.2023.e13535
3. Chibeba, A. M., Kyei-Boahen, S., Guimarães, M. de, Nogueira, M. A., & Hungria, M. (2018). Feasibility of transference of inoculation-related technologies: A case study of evaluation of soybean rhizobial strains under the agro-climatic conditions of Brazil and Mozambique. Agriculture, Ecosystems and Environment, (261), 230–240. https://doi.org/10.1016/j.agee.2017.06.037
4. Deaker, R., Roughley, R. J., & Kennedy, I. R. (2004). Legume Seed Inoculation Technology- a Review. Soil Biology and Biochemistry, 36(8), 1275–1288. https://doi.org/10.1016/j.soilbio.2004.04.009
5. Gelfand, I., & Philip Robertson, G. (2014). A reassessment of the contribution of soybean biological nitrogen fixation to reactive N in the environment. Biogeochemistry, 123(1–2), 175–184. https://doi.org/10.1007/s10533-014-0061-4
6. Herridge, D. F., Peoples, M. B., & Boddey, R. M. (2008). Global inputs of biological nitrogen fixation in Agricultural Systems. Plant and Soil, 311(1–2), 1–18. https://doi.org/10.1007/s11104-008-9668-3
7. Li, Y., Shen, Y., Shi, R., Yang, Z., Chen, Y., Luo, W., Wei, G., & Chou, M. (2023). The synthesis and secretion of key substances in the flavonoid metabolic pathway responding to different nitrogen sources during early growth stages in Robinia pseudoacacia. Plant and Soil, 494(1–2), 373–393. https://doi.org/10.1007/s11104-023-06286-y
8. Pudasaini, R. (2024). White fonio phenotyping and legume nodule crushing to assist smallholder farmers [Doctoral dissertation, University of Guelph]. https://atrium.lib.uoguelph.ca/items/20e80e87-f60f-4c34-8da2-17131e85c2c8
9. Pudasaini, R., & Raizada, M. N. (2024). Nodule crushing: A novel technique to decentralize rhizobia inoculant technology and empower small-scale farmers to enhance legume production and income. Frontiers in Sustainable Food Systems, 8(1423997). https://doi.org/10.3389/fsufs.2024.1423997
10. Pudasaini, R., Hewedy, O. A., & Raizada, M. N. (2023). Improving field legume nodulation by crushing nodules onto seeds: Implications for small-scale farmers. Frontiers in Agronomy, 5, 1161978. https://doi.org/10.3389/fagro.2023.1161978
11. Wang, Q., Liu, J., & Zhu, H. (2018). Genetic and molecular mechanisms underlying symbiotic specificity in legume-rhizobium interactions. Frontiers in Plant Science, 9, 00313. https://doi.org/10.3389/fpls.2018.00313